Abstract
Autistic people experience a disproportionately high rate of sleep disturbance, GI dysfunction, food sensitivity, sedentary behavior, metabolic dysregulation, and chronic stress. Despite these well-documented risks, intervention models to date have prioritized behavioral and developmental engagement over long-term health improvements. The very concept of healthspan, the period of life spent in good health, provides a complementary framework that emphasizes prevention, restoring functional capacity, and lifestyle interventions. This narrative review examines six core healthspan domains: exercise, sleep, nutrition, gut health, metabolic health, and stress regulation, and evaluates how these domains might be adapted to meet autism-specific requirements. A narrative review informed by a systematic search was conducted across several major databases, with 32 studies included in the final synthesis. Evidence suggests that these domains are both disrupted and modifiable in autistic populations. Emerging research on the gut-brain axis highlights potential biomechanical links, though clinical applications are still in the early stages. Across the domains, intervention effectiveness is shaped by individual sensory sensitivities, interoceptive differences, executive functioning challenges, and caregiver context. Autism-adapted healthspan interventions may be associated with improvements in functioning, quality of life, and long-term health outcomes, representing a promising and underutilized opportunity for integrative and preventative autism care.
Keywords
autism spectrum disorder healthspan lifestyle intervention sleep nutrition physical activity gut microbiome
1. Introduction
Autism spectrum disorder (ASD) is a lifelong neurodevelopmental condition characterized by differences in social communication and the presence of restricted or repetitive behaviors. Although traditionally conceptualized through a developmental and behavioral lens, there has been increasing recognition that autism is also associated with significant and persistent physical health disparities across the lifespan. Autistic individuals experience elevated rates of chronic conditions, including sleep disorders, gastrointestinal dysfunction, metabolic abnormalities, and mental health challenges ([15]; [13]).
Historically, intervention efforts in autism have focused primarily on behavioral therapies, educational supports, and, in some cases, pharmacologic management of co- occurring symptoms. While these approaches are often essential, in many cases, they fail to address broader determinants of health that influence long-term outcomes. Autistic individuals experience the cumulative impact of lifestyle-related factors, including physical inactivity, sleep disruption, nutritional limitations, and chronic stress. These factors may contribute to increased morbidity and reduce the quality of life within the autistic community.
The concept of healthspan, defined as the duration of life spent in good health, provides a valuable framework for addressing these challenges. Healthspan-oriented approaches emphasize prevention, functional optimization, and the integration of lifestyle-based interventions. In recent years, there has been a growing interest in applying principles of lifestyle medicine to neurodevelopmental conditions, including autism.
Emerging evidence suggests that key healthspan domains, including exercise, sleep, nutrition, gut health, metabolic regulation, and stress management, are both disruptive and modifiable in autistic populations. However, effective implementation requires careful adaptation to autism-specific characteristics, including sensory sensitivities, interoceptive differences, executive functioning challenges, and reliance on structured environments.
This review synthesizes current evidence across these domains and proposes that autism- adapted healthspan interventions may be associated with improvements. These considerations highlight the need for integrative frameworks that extend beyond traditional intervention models.
2. Methods
2.1 Study Design
This study was conducted as a narrative review informed by systematic search principles. While elements of PRISMA were used to guide transparency in study identification and selection, this review did not include formal risk-of-bias assessment or meta-analysis and therefore should not be considered a full systematic review. A narrative synthesis approach was selected due to heterogeneity in study designs, intervention types, and outcome measures.
2.2 Search Strategy
A structured literature search was conducted across PubMed/MEDLINE, Scopus, PsycINFO, and Google Scholar. Studies published between January 2000 and February 2026 were considered for inclusion. Search terms included combinations of “autism,” “autism spectrum disorder,” “exercise,” “sleep,” “nutrition,” “gut microbiome,” “metabolic,” “stress,” and “lifestyle interventions.” Boolean operators and database- specific indexing terms were applied.
2.3 Inclusion and Exclusion Criteria
Included studies were peer-reviewed articles, including systematic reviews, meta- analyses, randomized controlled trials, and observational studies involving autistic individuals across the lifespan. Studies were required to address at least one healthspan domain. Exclusion criteria included non-peer-reviewed publications, case reports without generalizability, non-English studies, and pharmacologic-only interventions without lifestyle relevance.
2.4 Study Selection
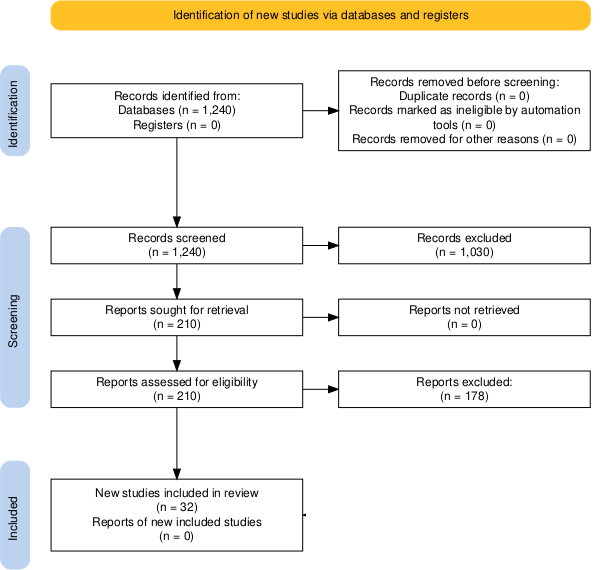
Approximately 1,240 records were identified during this research. Following a title and abstract screening, 210 articles underwent full-text review. Of these 210 articles, 68 met the eligibility criteria, and ultimately, 32 studies were included in the final synthesis.
Screening and selection were conducted by a single reviewer. The study selection process is summarized in a PRISMA 2020-style flow diagram (Figure 1).
2.5 Data Synthesis
The data in this research were extracted qualitatively and synthesized across six domains: exercise, sleep, nutrition, gut health, metabolic health, and stress regulation. Study selection procedures were documented using a PRISMA-style framework to enhance transparency. No quantitative meta-analysis was performed due to heterogeneity across study designs and outcome measures.
3. Conceptual Framework: Autism-Adapted Healthspan Model
Healthspan interventions are designed to optimize physiological systems and reduce chronic disease risk through lifestyle adjustment. In this context, the present review proposes a conceptual model for autism-adapted healthspan intervention (AAHI model), in which core lifestyle domains are systematically adapted to align with the neurobiological, sensory, and behavioral characteristics of autistic individuals. The structure of the AAHI model is illustrated in Figure 2. Within the AAHI model, intervention effectiveness may be influenced by four core autism-specific modifiers: sensory processing differences, interoceptive variability, executive functioning challenges, and preferences for sameness and predictability.
Autistic individuals frequently exhibit sensory processing differences that can affect tolerance for environmental stimuli. These stimuli may include noise, light, smell, and tactile inputs. Variability in the perception of internal body signals, known as interoception, may influence perceptions of hunger, fatigue, and emotional awareness in this population. Executive functioning challenges can also affect planning, initiation, and adherence to routines, while a preference for sameness may both support and constrain behavior change.
These factors suggest that standard lifestyle interventions may not translate directly to an autistic population without modification. Instead, interventions must be individualized, structured, and context-sensitive, emphasizing predictability, sensory compatibility, and gradual adaptation. This framework aligns with emerging models in precision, holisticlifestyle medicine. This structure forms the basis of the AAHI model, which is further elaborated across the following domains.
In summary, the AAHI model consists of three interacting components: (1) six core healthspan domains, (2) four autism-specific modifiers that influence intervention design and response, and (3) an implementation context that determines feasibility and sustainability. Together, these components define the AAHI model as a structured framework for developing individualized, autism-adapted lifestyle interventions.
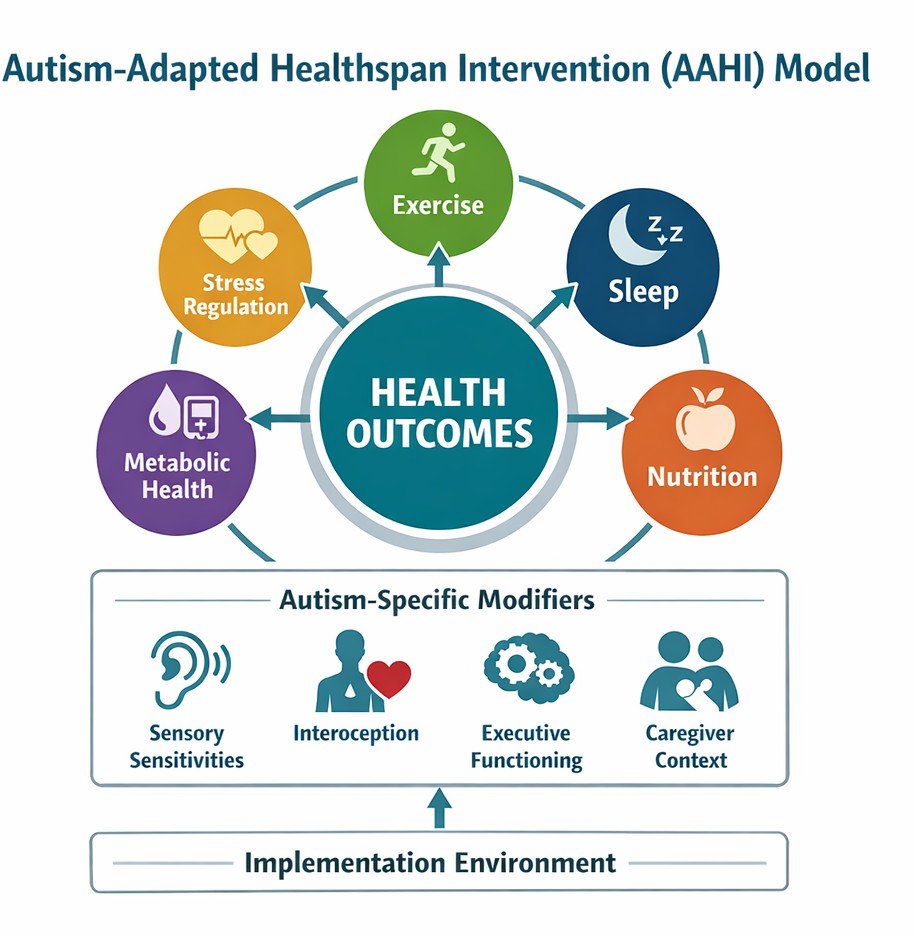
The model depicts the interaction between core healthspan domains, autism-specific modifiers, and implementation context in shaping health outcomes.
4. Exercise and Physical Activity
Physical activity is a foundational component of healthspan, contributing to cardiovascular health, metabolic regulation, and psychological well-being. Autistic individuals, however, consistently demonstrate lower levels of physical activity and higher levels of sedentary behavior compared to neurotypical peers [9].
Exercise interventions have been associated with improvements in both physical and behavioral outcomes in autistic populations. Systematic reviews and meta-analyses suggest that structured physical activity may reduce stereotypical behaviors, improve social engagement, and enhance overall fitness ([2]; [18]). The benefits of exercise may be mediated through neurobiological mechanisms, including increased neurotransmitter regulation and improved sensory integration.
Effective implementation of an exercise program in an autistic population requires adaptation. Sensory sensitivities may limit participation in traditional gym or team sport environments. As such, low-stimulation, predictable activities, like walking, swimming, or cycling, may be more accessible. Structured routines, visual supports, and individualized pacing can further enhance engagement.
Importantly, consistency and personal preference appear to be more critical determinants of success than the intensity of the exercise. Collectively, these findings suggest that adapted physical activity interventions may contribute to improvement in both physiological health and behavioral functioning.
5. Sleep and Circadian Regulation
Sleep disturbances are among the most prevalent co-occurring conditions in autism. These disturbances affect a substantial proportion of individuals and contribute to behavioral, cognitive, and emotional challenges [4]. Common issues include delayed sleep onset, reduced sleep duration, and irregular sleep-wake patterns.
Sleep plays a central role in healthspan. Sleep influences metabolic regulation, immune functions, and neuroplasticity. In autism, sleep disruption may exacerbate existing challenges, creating a bidirectional relationship between sleep and behavior.
Pharmacological interventions, particularly melatonin supplementation, have demonstrated efficacy in improving sleep onset and duration [6]. However, behavioral and environmental strategies are equally important. These include establishing consistent routines, optimizing the sleep environment to reduce sensory inputs, and supporting functional transitions to the sleep state.
Autism-adapted sleep interventions emphasize predictability, sensory compatibility, and caregiver involvement. Improvements in sleep may have cascading effects across other domains, including behavior, mood, and metabolic health.
6. Nutrition and Feeding Behavior
Nutrition is another critical, often overlooked, domain in autism. Food selectivity, characterized by limited dietary variety and strong preferences, is highly prevalent and may lead to nutritional deficiencies [12]. Sensory sensitivities to texture, taste, and appearance often contribute to these behavior patterns.
Studies indicate that autistic individuals are more likely to consume restricted diets and exhibit food refusal behaviors [1]. These patterns may increase the risk of both micronutrient deficiencies and imbalanced macronutrient intake. Interventions targeting feeding behavior often involve behavioral strategies, including gradual exposure to new foods and reinforcement techniques [16]. However, traditional approaches may be limited if they do not adequately address underlying sensory factors. Autism-adapted nutrition interventions prioritize reducing sensory distress, supporting autonomy, and gradually expanding dietary variety to improve nutritional well-being.
7. Gut Health and the Gut–Brain Axis
Gastrointestinal (GI) symptoms, including constipation, diarrhea, and abdominal pain, are more prevalent in the autistic population and are associated with behavioral challenges [8]. Increasing attention is being focused on the gut-brain axis, a bidirectional communication system linking gastrointestinal and neurological function. Emergent research suggests that alterations in the gut microbiome may influence behavior through immune, metabolic, and neural pathways. Experimental studies have demonstrated that microbiota derived from autistic individuals can influence behavior in animal models [17].
Clinical interventions targeting the microbiome, including microbiota transfer therapy, have demonstrated preliminary promise in early-stage studies, though findings remain inconsistent and require replication [10]. While the gut-brain axis represents a compelling area of research, current evidence supports cautious interpretation. Further studies are needed to establish clinical efficacy and long-term safety.
8. Metabolic Health and Long-Term Risk
Metabolic health is an emerging area of concern in the autistic population. Evidence suggests increased prevalence of both underweight and obesity, as well as elevated risk for cardiometabolic conditions ([11]; [5]). These risks may be influenced by physical inactivity, sleep disruption, and dietary limitations.
Metabolic dysfunction has significant implications for long-term healthspan, contributing to increased risk of cardiovascular disease, diabetes, and reduced life expectancy. In autistic populations, these risks may be compounded by barriers to healthcare access and health-promoting behaviors.
Integrating lifestyle interventions that address multiple domains simultaneously, such as physical activity, sleep, and nutrition, may offer a promising approach to improving metabolic outcomes. Early intervention may be particularly important in mitigating long- term risk.
9. Stress Regulation and Mental Health
Chronic stress and anxiety are common in autism and may significantly impact the quality of life. Factors contributing to stress include sensory overload, social demands, and environmental unpredictability. Mindfulness-based interventions have demonstrated effectiveness in providing emotional regulation and reducing anxiety in autistic individuals ([19]; [3]). These interventions may support greater awareness and regulation of internal states. Adaptations are often necessary to ensure accessibility, including structured formats, concrete language, and incorporation of movement-based practices. Stress regulation strategies may also have indirect effects in other domains, including sleep and behavior.
9. Caregiver and Environmental Context
Caregivers play a central role in implementing and sustaining health interventions in autism. Parenting stress is significantly elevated and may influence both child intervention success ([7]; [14]). Family-centered approaches that consider caregiver capacity, environmental context, and resource availability are essential. Supporting caregivers may enhance the effectiveness and sustainability of healthspan interventions. The AAHI model further emphasizes the role of implementation context, including caregiver capacity, environmental structure, and resource availability, as critical determinants of intervention feasibility and sustainability.
11. Integration: Toward an Autism-Adapted Healthspan Model
Healthspan domains are highly interconnected, with changes in one domain often influencing others. For example, improvements in sleep may enhance metabolic regulation and reduce stress, while increasing physical activity may support both mental health and sleep quality. The AAHI model conceptualizes healthspan outcomes as emerging from the dynamic interaction between core lifestyle domains, autism-specific modifiers, and implementation context. This approach reflects a shift toward systems- based, longitudinal care that integrates behavioral, physiological, and environmental factors. Taken together, these findings support the AAHI model, which emphasizes integrated, multi-domain, and individualized healthspan interventions tailored to autistic individuals. This integrative perspective positions the AAHI model as a potential framework for guiding both clinical application and future research.
12. Limitations
This review is limited by the heterogeneity of the included studies, variability in intervention design, and limited longitudinal data. Additionally, the narrative design does not allow for quantitative synthesis, and publication bias and variability in outcome measures may limit generalizability.
13. Conclusion
Autistic individuals experience complex and interrelated health challenges that extend beyond common diagnostic conditions. Healthspan-oriented interventions offer a promising framework for addressing these challenges through integrated lifestyle-based approaches. Future research should prioritize the development and evaluation of integrated, autism-adapted, lifestyle interventions to support health across the lifespan.
References
- Bandini, L. G., Anderson, S. E., Curtin, C., Cermak, S., Evans, E. W., Scampini, R., Maslin, M., & Must, A. (2010). Food selectivity in children with autism spectrum disorders and typically developing children. The Journal of Pediatrics, 157(2), 259–264. DOI ↗ Google Scholar ↗
- Bremer, E., Crozier, M., & Lloyd, M. (2016). A systematic review of the behavioral outcomes following exercise interventions for children and youth with autism spectrum disorder. Autism, 20(8), 899–915. DOI ↗ Google Scholar ↗
- Cachia, R. L., Anderson, A., & Moore, D. W. (2016). Mindfulness interventions for individuals with autism spectrum disorder: A systematic review and meta- analysis. Journal of Child and Family Studies, 25, 1–14. DOI ↗ Google Scholar ↗
- Carmassi, C., Palagini, L., Caruso, D., Masci, I., Nobili, L., Vita, A., & Dell’Osso, L. (2019). Systematic review of sleep disturbances and circadian sleep desynchronization in autism spectrum disorder. Journal of Clinical Medicine, 8(4), 486. DOI ↗ Google Scholar ↗
- Dhanasekara, C. S., Sadasivam, B., & Ramachandran, S. (2023). Association between autism spectrum disorder and cardiometabolic diseases: A systematic review and meta-analysis. Autism Research, 16(2), 230–245. DOI ↗ Google Scholar ↗
- Gringras, P., Nir, T., Breddy, J., Frydman-Marom, A., & Findling, R. L. (2017). Efficacy and safety of pediatric prolonged-release melatonin for insomnia in children with autism spectrum disorder. Journal of the American Academy of Child & Adolescent Psychiatry, 56(11), 948–957. DOI ↗ Google Scholar ↗
- Hayes, S. A., & Watson, S. L. (2013). The impact of parenting stress: A meta-analysis of studies comparing the experience of parenting stress in parents of children with and without autism spectrum disorder. Journal of Autism and Developmental Disorders, 43(3), 629–642. DOI ↗ Google Scholar ↗
- Hsiao, E. Y. (2014). Gastrointestinal issues in autism spectrum disorder. Harvard Review of Psychiatry, 22(2), 104–111. DOI ↗ Google Scholar ↗
- Jones, R. A., Downing, K., Rinehart, N. J., Barnett, L. M., May, T., McGillivray, J., & Hinkley, T. (2017). Physical activity, sedentary behavior and their correlates in children with autism spectrum disorder: A systematic review. PLoS ONE, 12(2), e0172482. DOI ↗ Google Scholar ↗
- Kang, D. W., Adams, J. B., Gregory, A. C., Borody, T., Chittick, L., Fasano, A., Khoruts, A., Geis, E., Maldonado, J., McDonough-Means, S., Pollard, E. L., Roux, S., Sadowsky, M. J., & Krajmalnik-Brown, R. (2017). Microbiota transfer therapy alters gut ecosystem and improves gastrointestinal and autism symptoms. Microbiome, 5(1), 10. DOI ↗ Google Scholar ↗
- Li, Y. J., Xie, X. N., Lei, X., Zhang, Y. Y., Gao, Y. T., & Li, H. H. (2020). Global prevalence of obesity, overweight and underweight in children, adolescents and adults with autism spectrum disorder: A systematic review and meta-analysis. Obesity Reviews, 21(12), e13103. DOI ↗ Google Scholar ↗
- Marí-Bauset, S., Zazpe, I., Mari-Sanchis, A., Llopis-González, A., & Morales-Suárez- Varela, M. (2014). Food selectivity in autism spectrum disorders: A systematic review. Journal of Child Neurology, 29(11), 1554–1561. DOI ↗ Google Scholar ↗
- Micai, M., Fulceri, F., Caruso, A., & Muratori, F. (2023). Co-occurring conditions in autism spectrum disorder: A systematic review. Journal of Clinical Medicine, 12(3), 1124. DOI ↗ Google Scholar ↗
- Neece, C. L., Green, S. A., & Baker, B. L. (2012). Parenting stress and child behavior problems: A transactional relationship across time. American Journal on Intellectual and Developmental Disabilities, 117(1), 48–66. DOI ↗ Google Scholar ↗
- Rydzewska, E., Hughes-McCormack, L. A., Gillberg, C., Henderson, A., MacIntyre, C., Rintoul, J., & Cooper, S. A. (2018). Prevalence of long-term health conditions in adults with autism: Observational study of a whole country population. BMJ Open, 8(8), e023945. DOI ↗ Google Scholar ↗
- Sharp, W. G., Jaquess, D. L., Morton, J. F., & Herzinger, C. V. (2013). Pediatric feeding disorders: A quantitative synthesis of treatment outcomes. Clinical Child and Family Psychology Review, 16(4), 348–365. DOI ↗ Google Scholar ↗
- Sharon, G., Cruz, N. J., Kang, D. W., Gandal, M. J., Wang, B., Kim, Y. M., Zink, E. M.,Casey, C. P., Taylor, B. C., Lane, C. J., Bramer, L. M., Isern, N. G., Hoyt, D. W., Noecker, C., Sweredoski, M. J., Moradian, A., Borenstein, E., Jansson, J. K., Knight, R., & Mazmanian, S. K. (2019). Human gut microbiota from autism spectrum disorder promote behavioral symptoms in mice. Cell, 177(6), 1600– 1618. DOI ↗ Google Scholar ↗
- Sowa, M., & Meulenbroek, R. (2012). Effects of physical exercise on autism spectrum disorders: A meta-analysis. Research in Autism Spectrum Disorders, 6(1), 46–57. DOI ↗ Google Scholar ↗
- Spek, A. A., van Ham, N. C., & Nyklíček, I. (2013). Mindfulness-based therapy in adults with an autism spectrum disorder: A randomized controlled trial. Research in Developmental Disabilities, 34(1), 246–253. DOI ↗ Google Scholar ↗